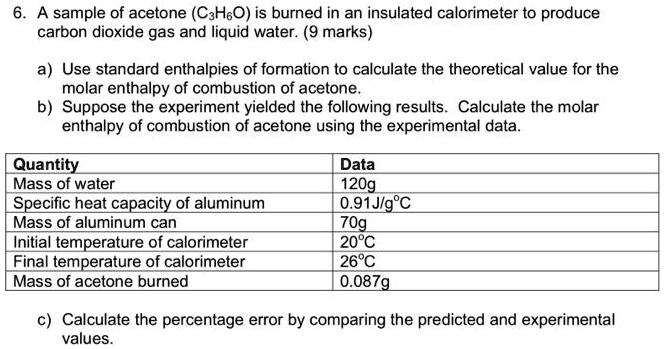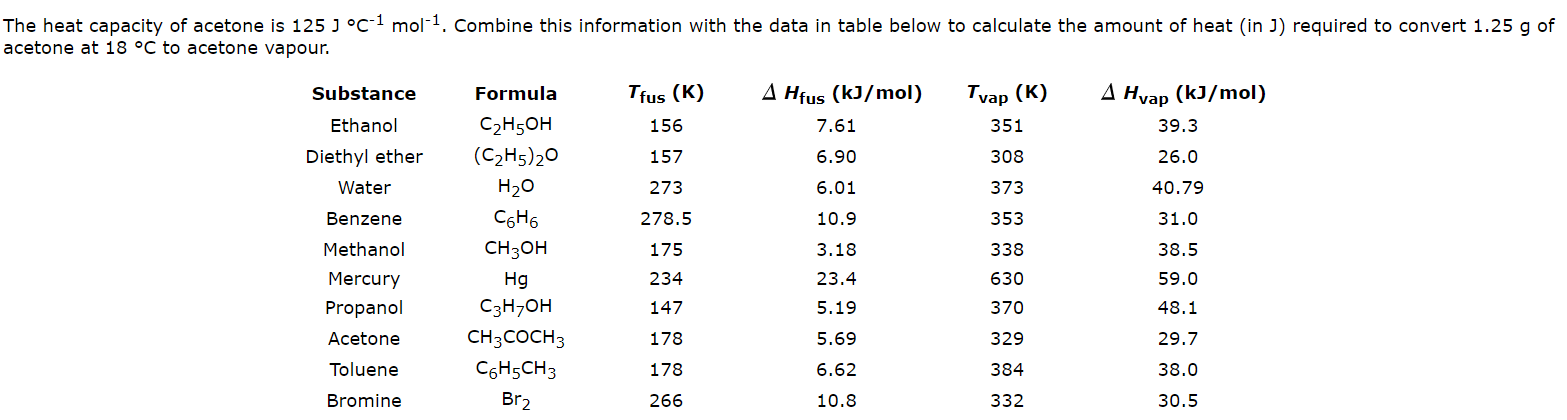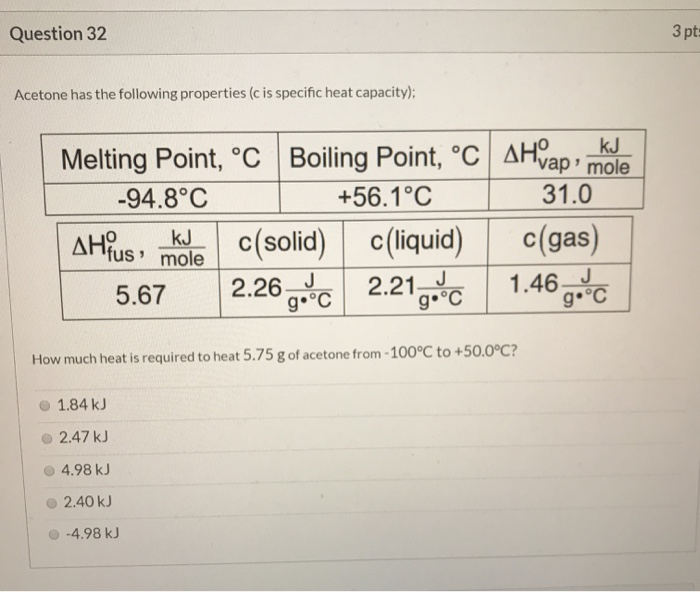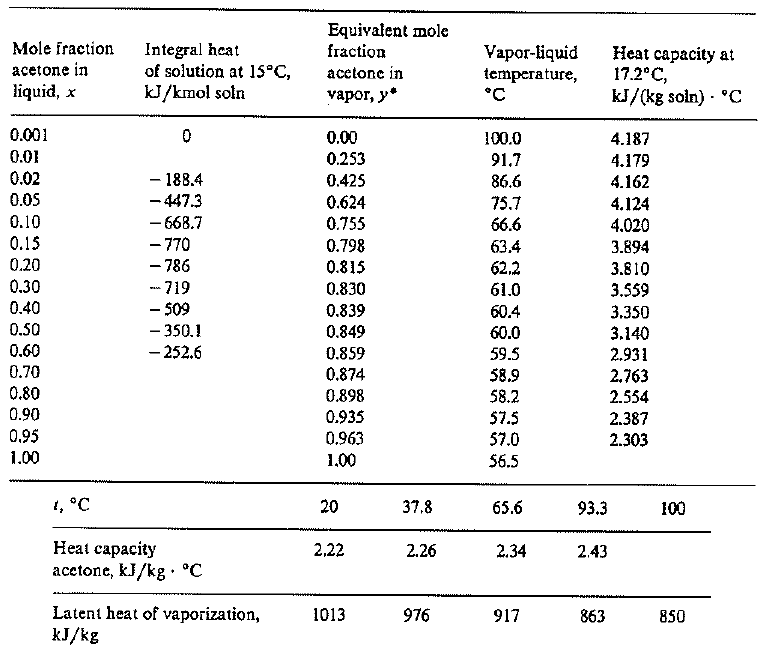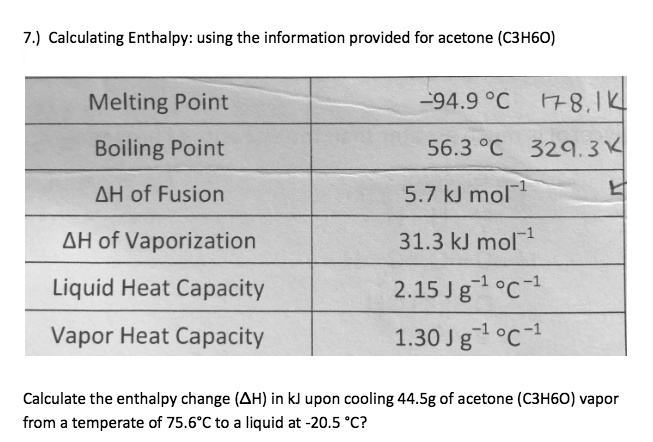
SOLVED: 7.) Calculating Enthalpy: using the information provided for acetone (C3HGO) Melting Point Boiling Point AH of Fusion 94.9 %€ +7-8,1444 56.3 "C 329.34 5.7 kJ mol-1 AH of Vaporization 31.3 kJ

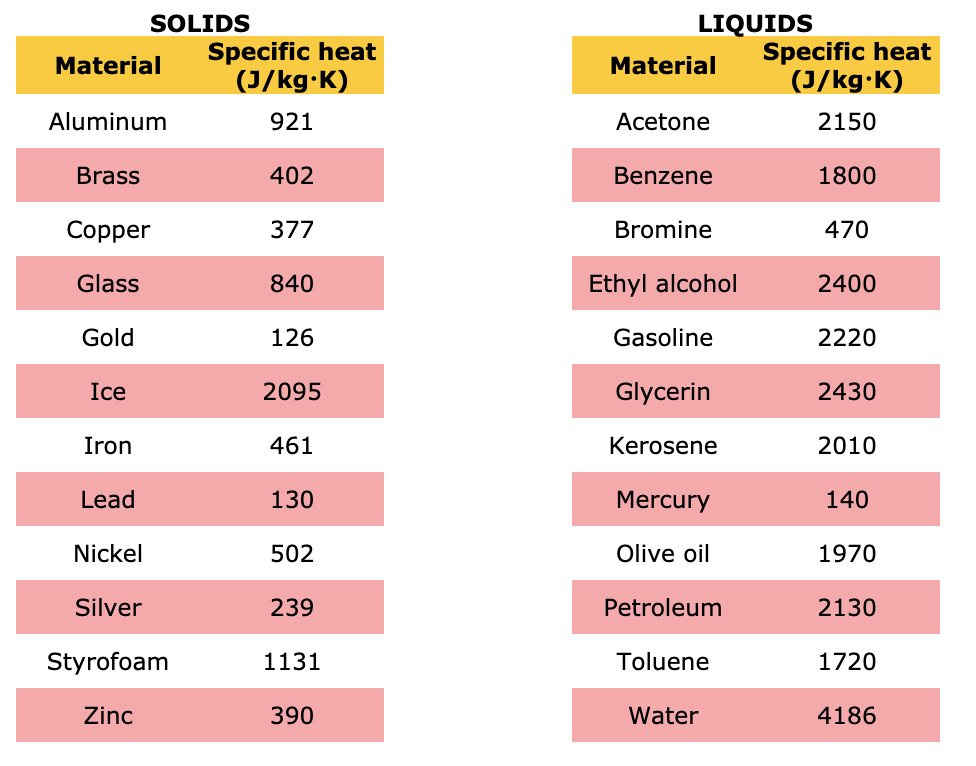
SOLVED: Questions: Which of the following metals is 0.91 JIg"C or cast iron with better conductor Of heat; Al with specific heat of specific heal of 46 JlgPc? The specific heat of

SOLVED: A solid substance has a mass 0f 250.00g It is cooled by 25.00*C and loses 4.937kJ of heat What is the specific heat capacity of the substance in 9.PC [APP 2

Heat of vaporization of acetone. Simulation data: • this work, AUA4... | Download Scientific Diagram

Using the given data and calculate the enthalpy of formation of acetone(g). Bond enthalpy of : C - H = 415 ; C - C = 350 ; (C = O) =

Thermophysical Properties of Liquid Acetone, Cellulose Acetate, and... | Download Scientific Diagram